Purification Made Simple
Clinical Manufacturing Is The Bottleneck Of Drug Development
Clinical manufacturing is the bottleneck of drug development. LetsGo 3.1 is a turnkey solution with shorter delivery timeline and one third of cost
2/20/20263 min read
The Real Cost of Time in Drug Development
Drug development is often described as expensive. What’s less discussed is how much of that cost is driven by time. Every delay in the pipeline compounds spending, ties up capital, and postpones patient access.
Clinical trials and clinical manufacturing sit at the center of this challenge. They are essential, tightly regulated, and resource-intensive. But they are also among the most time-consuming and capital-heavy stages in the entire development lifecycle.
Clinical Trials: Long Timelines by Design
Clinical trials are complex by necessity. Before a single patient is dosed, sponsors must complete:
Preclinical studies and toxicology packages
IND preparation and regulatory submissions
Site selection and contracting
Patient recruitment planning
Supply chain coordination
Once trials begin, timelines stretch further. Phase I, II, and III studies often span years. Recruitment delays, protocol amendments, and safety monitoring can extend schedules even more.
Each month of delay has financial consequences. Development teams remain staffed. CRO contracts continue. Trial sites remain active. Capital remains committed without revenue.
For publicly traded companies, extended timelines also impact valuation and investor confidence. For private biotech firms, delays can shorten runway and force additional fundraising under pressure.
Time in clinical development is not neutral. It is expensive.
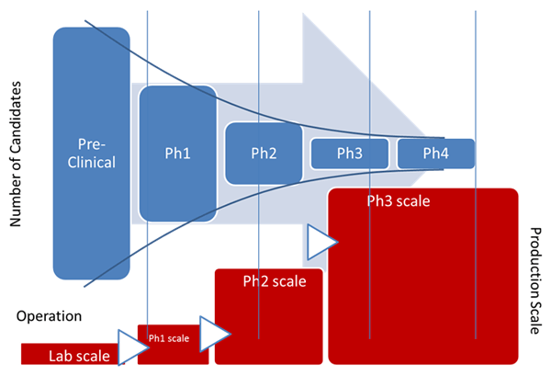
Clinical Manufacturing: The Hidden Bottleneck
While clinical trial execution is visible, clinical manufacturing is often the less visible constraint that shapes timelines behind the scenes.
Producing clinical-grade drug substance and drug product requires:
Process development and scale-up
GMP facility access or construction
Equipment procurement and installation
Validation and qualification activities
Raw material sourcing and quality assurance
If manufacturing capacity is not available when trials are ready to begin, programs wait.
In many cases, building or expanding GMP manufacturing infrastructure can take two to three years. That includes facility design, construction, cleanroom certification, equipment installation, commissioning, and validation. These efforts require specialized engineering teams, quality oversight, and regulatory alignment.
The capital required is substantial. Clinical manufacturing facilities can demand hundreds of millions of dollars in investment, depending on scale and modality. Even outsourcing to CDMOs comes at a premium, particularly when capacity is constrained across the industry.
Manufacturing is not just a cost center. It is a timing gate.
Capital Intensity Across the Pipeline
The combined effect of long trials and heavy manufacturing investment creates a difficult dynamic:
Large capital outlays occur before proof of commercial success
Infrastructure is often committed early to avoid delays later
Financial risk accumulates while clinical uncertainty remains high
For emerging biotech companies, this can mean raising significant funding long before a product reaches late-stage trials. For larger pharmaceutical companies, it means allocating capital years in advance of potential revenue.
In both cases, the pipeline becomes a balance between speed, risk, and financial exposure.
Operational Complexity Multiplies Cost
Beyond direct capital expenditure, operational complexity adds further burden:
Maintaining GMP compliance requires extensive documentation and quality systems
Technology transfer between development and manufacturing teams consumes time and resources
Changes in clinical demand can require manufacturing adjustments or new capacity
Every transition point in the pipeline introduces risk of delay. Every delay increases cost.
Why Time and Capital Are So Closely Linked
In drug development, time is not simply a schedule metric. It directly translates into:
Ongoing payroll and operational expenses
Continued clinical site costs
Extended capital lock-up
Delayed market entry
Reduced effective patent life
The longer a program takes to reach commercialization, the shorter the revenue window under patent protection. That compression increases pressure on pricing and return on investment.
This is why development efficiency is not only a scientific concern but a strategic one.
The Strategic Imperative
Clinical trials and clinical manufacturing will always require rigor, compliance, and investment. Those fundamentals cannot be removed. But the industry continues to look for ways to reduce unnecessary delay, improve capital efficiency, and better align manufacturing readiness with clinical milestones.
Organizations that manage time effectively in the pipeline gain more than cost savings. They gain flexibility. They reduce risk. And they increase the likelihood that promising therapies reach patients before capital or momentum runs out.
In modern drug development, managing time is managing value.