Purification Made Simple
Is It Time to Platform Clinical Manufacturing in Biopharmaceutical Industry?
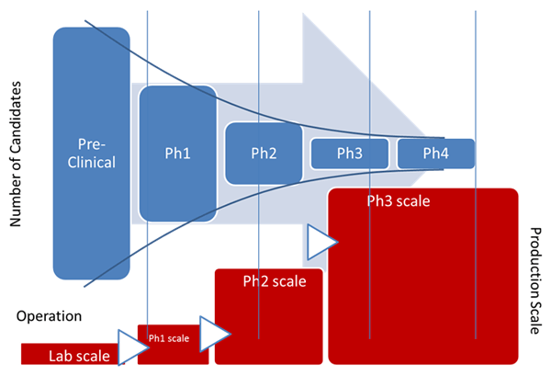
Clinical manufacturing has always been the bottleneck for drug development. With the progresses on modular system design and continuous processing, it becomes interestingly feasible that a configurable and re-deployable clinical manufacturing platform may be the future direction.
4/1/20263 min read
Is It Time to Platform Clinical Manufacturing?
For years, biopharmaceutical development has pushed toward greater speed and flexibility. Discovery cycles have accelerated. Clinical trial designs have become more adaptive. Data flows faster than ever.
But one part of the pipeline has struggled to keep up: clinical manufacturing.
Despite advances in science, manufacturing is still often built from scratch for each program. Facilities take years to design and construct. Processes are reconfigured repeatedly. Capacity decisions are made early, often before clinical outcomes are clear.
This raises a simple question:
Is it time to move clinical manufacturing onto a platform model?
The Problem with the Current Model
Traditional clinical manufacturing is highly customized. Each new molecule can trigger:
New facility builds or major modifications
Custom engineering design
Long validation cycles
Significant upfront capital investment
This approach worked when pipelines were smaller and timelines were longer. Today, it creates friction.
Programs wait for manufacturing readiness. Capital is committed too early. Scaling decisions are made with limited data. And when priorities shift, infrastructure does not adapt easily.
Manufacturing becomes a constraint instead of an enabler.
What a Platform Approach Changes
A platform model treats manufacturing not as a one-off build, but as a standardized, configurable system.
Instead of designing around each molecule, you deploy a flexible production platform that can be adapted through configuration rather than reconstruction.
At a practical level, this means:
Standard modules for key unit operations
Configurable process flow without redesign
Pre-defined control architecture
Repeatable validation framework
The goal is not to eliminate customization entirely, but to contain it within a structured system.
For engineers, this reduces integration complexity.
For decision-makers, it reduces time and capital risk.
Why Now?
Several industry shifts make platforming clinical manufacturing more viable today than before:
Increased Pipeline Pressure
More biologics, more modalities, and more parallel programs require faster manufacturing readiness.
Demand for Flexibility
Clinical outcomes are uncertain. Manufacturing capacity needs to scale up, down, or shift across programs.
Maturity of Continuous Processing
Continuous downstream technologies have reached a point where they can support stable, production-ready operation.
Advances in Automation and Control
Modern DCS systems and standardized batch frameworks (such as S88) enable consistent execution across deployments.
Together, these trends support a move away from facility-centric models toward deployable systems.
What a Clinical Manufacturing Platform Should Deliver
For a platform approach to work, it must go beyond standardization. It must also address real operational constraints.
A viable platform should be:
Modular and Configurable
Built from standardized modules that can be arranged for different downstream processes without redesign.
cGMP Compliant by Design
Compliance should be embedded in the system architecture, not added later.
Fast to Deploy
Deployment timelines should be measured in months, not years.
Minimal Construction Required
The system should reduce dependence on large facility builds, utilities, and infrastructure.
Re-deployable
Capacity should not be fixed in one location. Systems should be movable and reusable across programs.
Scalable Without Reinvention
Expansion should happen through replication or parallelization, not redesign.
These requirements define what a true manufacturing platform looks like in practice.
From Concept to Reality: Platforms Like LetsGo 3.1
This is where solutions such as LetsGo 3.1, developed by Lisure, come into focus.
LetsGo 3.1 represents a shift from theory to implementation. It delivers:
A module-based downstream system configurable for different DSP unit operations
A fully enclosed, cGMP-compliant production line
Continuous processing capability with steady-state operation
Minimal site construction requirements, with no need for extensive utilities such as CIP
Deployment timelines of around six months
A re-deployable platform that can be used across programs and sites
Instead of building manufacturing capability into a facility, the capability is delivered as a system.
This aligns manufacturing more closely with how clinical development actually works: dynamic, uncertain, and fast-moving.
What This Means for the Industry
Platforming clinical manufacturing changes more than engineering workflows. It changes strategy.
Time to clinic shortens because manufacturing no longer gates progress
Capital efficiency improves because infrastructure is not overbuilt
Risk decreases because investment aligns with program milestones
Operational consistency increases through standardized systems
Perhaps most importantly, manufacturing becomes adaptable.
In a pipeline where priorities shift and timelines compress, adaptability is as valuable as capacity.
So, Is It Time?
The idea of platforming clinical manufacturing is not new. What has changed is the ability to execute it.
With modular design, continuous processing, and turnkey deployment now available in integrated systems, the barriers that once slowed adoption are being removed.
The question is no longer whether platforming is possible.
It is whether continuing to build one-off manufacturing systems still makes sense in a world that demands speed, flexibility, and control.
For many organizations, the answer is becoming clear