Purification Made Simple
How LetsGo 3.1 Solves the Continuous Manufacturing (CM) Challenges
2/22/20263 min read
Continuous Manufacturing Challenges
Industry leaders were already calling continuous biomanufacturing a “changing processing paradigm.” The advantages were clear: better process control, smaller footprints, higher productivity, and improved quality consistency. Regulators were supportive. Case studies showed promise.
And yet, adoption was slow.
The challenges identified are real: Downstream technology immaturity, Integration complexity, Batch definition ambiguity, High upfront investment, Limited cGMP-ready equipment, Validation burden, and Organizational resistance.
Nearly a decade later, Lisure has introduced a solution designed specifically to close those gaps. LetsGo 3.1 is not a concept platform or pilot demonstration. It is a turnkey, continuous downstream clinical manufacturing system engineered to address, systemically, the barriers that slowed adoption in the past.
How LetsGo 3.1 resolves the core challenges?
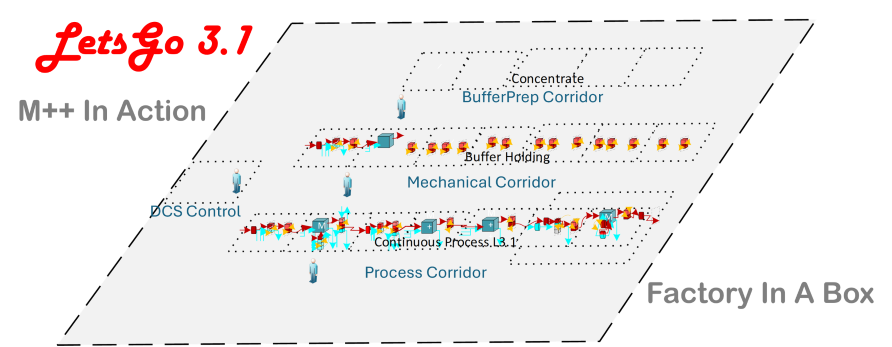
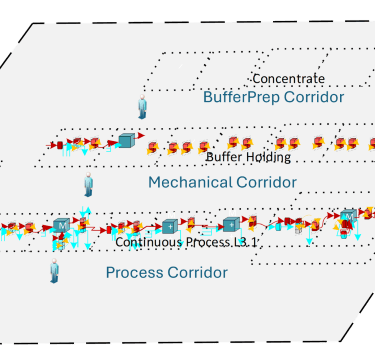
Modular, Production-Ready Architecture
Continuous downstream processing was considered technologically complicated and immature. Multi-column chromatography production systems have been maturing, but integration is still complex and cGMP-ready supply chains is limited.
LetsGo 3.1 is built on M++ modular standard architecture designed specifically for downstream DSP unit operations. Each module is engineered for continuous operation, integrated control, and cGMP deployment.
Instead of stitching together experimental components, LetsGo 3.1 delivers a unified, enclosed downstream production train:
Continuous capture and polishing
Inline dilution buffer preparation
Fully automated control
Integrated DCS and S88 batch management
Downstream continuous is no longer experimental. It is production-ready.
Batch Definition and Regulatory Consideration
How do you define a batch in continuous manufacturing? How do you ensure traceability during steady-state operation? Sampling and QA paradigms designed for batch processes did not directly translate.
LetsGo 3.1 is fully automated under DCS control with S88 batch management logic. Batch definition is structured through digital control architecture rather than physical hold tanks.
Key elements include:
Defined production windows
Continuous real-time monitoring
Structured electronic records
Full traceability across the steady-state production period (RTD based mini-batch)
By embedding control and traceability in the automation layer, LetsGo 3.1 aligns continuous operation with regulatory expectations for batch documentation and lot tracking readiness.
The paradigm shift from end-product testing to process dynamics, PAT, and off-line testing is operationalized, not theoretical.
Steady-State Continuous Execution
Continuous processing requires deep process understanding to maintain steady-state conditions. Traditional offline sampling was insufficient. Robust control systems were necessary.
LetsGo 3.1 is designed for constant feed-in and constant product-out operation. Continuous inline dilution eliminates large batch buffer prep variability. Control logic maintains required process parameters throughout the run.
Because the system translates existing batch processes into continuous mode without additional process redevelopment, it lowers the barrier to implementation while maintaining robust process control.
Steady-state operation is engineered into the hardware and automation from day one.
Fully Enclosed Factory-in-a-Box
Integration between upstream and downstream operations was a known risk. Unit operations interact dynamically, and retrofitting facilities created complexity.
LetsGo 3.1 is delivered as a fully enclosed, turnkey downstream production line. It replaces permanent infrastructure with a re-deployable platform.
Key integration advantages:
No CIP utilities required
Minimized site construction
Small equipment footprint
Standardized module interfaces
One-click automated operation
Rather than retrofitting legacy facilities, organizations deploy a contained production environment. This eliminates many of the integration uncertainties that slowed adoption in earlier years.
Accelerated Deployment Model
Continuous manufacturing required significant upfront investment in process development, facility adaptation, and validation. For clinical-stage products, this risk was hard to justify.
Deployment time for LetsGo 3.1 is approximately six months. Capital and operational investment is less than a third of that of traditional clinical manufacturing builds.
The system:
Requires minimal construction
Has no CIP utility demand
Is pre-engineered for cGMP operation
Supports copy-and-paste tech transfer
Validation is focused on a standardized platform rather than a custom-built facility. The financial risk profile shifts from infrastructure-heavy to deployment-focused.
This directly addresses the early-stage adoption hurdle.
Designed-for-Continuous Hardware
Questions were raised about whether existing stainless-steel and single-use systems were designed for constant operation. Wear, reliability, and long-term stability were concerns.
The platform of LetsGo 3.1 is engineered specifically for continuous operation from the smallest hardware component, not adapted from batch equipment. Reduced column size, optimized resin utilization (up to 50% reduction in Protein A), and smaller system components reduce mechanical and operational stress.
Parallel deployment capability allows redundancy and flexible scaling without overburdening single systems.
Continuous reliability is built into the design philosophy.
Simplified Execution Model
Shifting from batch to continuous required new skill sets, new control philosophies, and reallocation of human resources.
True one-click operation by LetsGo 3.1 removes many manual interventions. Automation reduces operator variability. Standardized architecture simplifies training across programs and sites.
Instead of requiring organizations to become continuous manufacturing experts from scratch, LetsGo 3.1 encapsulates that expertise within the platform.
Continuous processing becomes accessible, not intimidating.
Downstream as a Deployable Platform
End-to-end continuous manufacturing is aspirational but still rarely implemented.
LetsGo 3.1 provides a practical foundation by making downstream continuous manufacturing deployable today. For many biologics, downstream was the most immature and difficult component. By stabilizing and standardizing downstream, the pathway toward integrated continuous manufacturing becomes achievable.
Downstream processing is no longer the bottleneck.
Continuous Manufacturing (CM), Operationalized
The industry understands the benefits of continuous manufacturing but struggled with practical implementation.
LetsGo 3.1 represents the transition from discussion to implementation.
It addresses:
Technological immaturity
Regulatory uncertainty
Integration risk
Capital intensity
Validation burden
Organizational complexity
Not through incremental improvement, but through a fully enclosed, automated, modular, turnkey downstream production platform.
Continuous manufacturing is no longer a changing paradigm. With LetsGo 3.1, it is a deployable clinical manufacturing reality.